Diaphragm pumps are key components in medical equipment to fight COVID-19
An (Artificial) Heart for Children
For its ventricular assist device, Berlin Heart relies on continually reliable KNF diaphragm pumps with low-noise operation

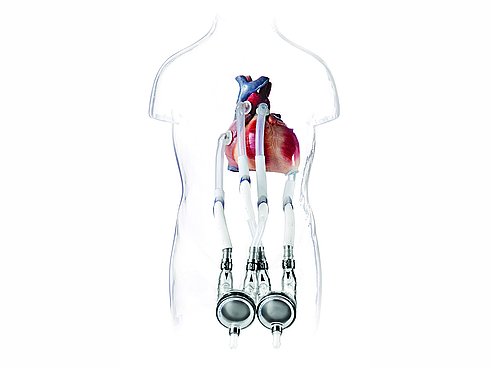
Our heart is a wonder of nature. For a healthy person, it steadily beats up to 100,000 times a day – with no rest. For cardiac patients urgently depending on a new heart, the situation is somewhat different. Their chance of survival depends crucially on finding a suitable donor heart. However, the waiting lists are long. A ventricular assist device (VAD) can help the patients on an interim basis. The EXCOR® Pediatric system from Berlin Heart is unique among these devices. It is the only ventricular assist device worldwide that has also been approved for children and babies. KNF provides highly specialized diaphragm gas pumps for the stationary drive unit.
From a start-up to the European market leader for ventricular assist devices
The international medical technology manufacturer Berlin Heart GmbH has its headquarters in Berlin and is a real Berlin story itself. It began in the late 80s in the form of a spin-off company of the German Heart Center in Berlin. In 1996, conceptualized by clinicians and developers and supported by Fehling Medical, Berlin Heart was founded in its present form. Since then, the company has become the European pioneer and market leader for ventricular assist devices and now employs more than 200 people.
Mechanical blood pump simulates cardiac function for people with cardiac insufficiency
The company’s core product is the EXCOR® blood pump, a double-chamber pump that takes over the cardiac functions for patients with cardiac insufficiency. The pump is placed on the outside of the patient’s body; cannulas connect it with the heart and the blood vessels. A pneumatic drive unit generates pressure and vacuum, which powers a three-layer diaphragm that separates the air chamber from the blood chamber, moving the membrane in a pulsating manner to create blood flow.
In the 90s, Berlin Heart developed the first heart pump for the treatment of children – a global breakthrough. For the related stationary drive unit Ikus, the company was looking for a reliable partner to develop a suitable compressor. In 1993, Berlin Heart found this partner – the diaphragm pump manufacturer KNF.
Controlled quality and reliability allow for a quick project implementation
As therapies may take months or even years, EXCOR® and all its components are designed for long-term use. Therefore, the medical technology manufacturer provided clear guidelines for the development of the KNF diaphragm gas pumps for the drive unit. In addition to performance and durability, the most important factor was low-noise operation to improve the patient’s quality of life during therapy. After reviewing all aspects of the project, the developers at Berlin Heart determined that the diaphragm pump technology from KNF could fulfill all requirements.
The collaboration benefited from the proven “modular KNF system” – a system that draws from pre-approved KNF pump components to facilitate speedy response and pump development. The pump was further adjusted to the individual project requirements from Berlin Heart. “Despite our special requirements, a complete redevelopment of the diaphragm pumps was not necessary”, said Daniel Hartmann, Team Leader Sustaining Engineering at Berlin Heart. “Instead, we could draw on the already proven and reliably tested components from the modular KNF system. That also complied with our wish to develop a reliable drive system within a short period of time.”
Together, Berlin Heart and the KNF development team optimized the parameters using the modular system. Starting with the existing series model “N 023.1.2”, this pump was perfectly adjusted to the customer requirements. Among other modifications, specially adapted intermediate plates and special silicon wiring were implemented. Due to the careful harmonization of all components, Berlin Heart's EXCOR® Pediatric product received premarket approval (PMA) for the US market.

Saving lives – the driver of business success
Since 1994, the EXCOR® Pediatric remains the only VAD approved for children, infants and babies and has saved more than 2,500 lives. “With each positive feedback on the successful use from the market, especially for children is always providing renewed strength and happiness, and this is exactly our main driver at Berlin Heart”, says Hartmann. Over the years, the company has opened up more and more markets. In 2005, the subsidiary Berlin Heart Inc. was founded in the USA where, in 2017, EXCOR® Pediatric also received premarket approval (PMA) from the FDA. In 2015, the first patients in Japan were successfully treated with the new system. Today, Asia is one of the biggest markets for the EXCOR® Pediatric artificial heart, and it has now been used successfully throughout the world.
Opening new markets requires continuous development
Opening new markets also changes the requirements for the heart pump. For example, the drive system's voltage needs to be changed depending on the country. Here, Berlin Heart could also rely on the KNF expertise. Through the years, KNF has continually developed the solution according to the new requirements. “We now offer five different voltages. This way, our system can be used almost anywhere in the world,” Hartmann explains. “KNF has considered our problems and interests throughout the project, and has proactively dealt with them. Furthermore, they always gave us the feeling of really appreciating our application. Still today, working with them is a big pleasure!”